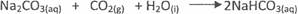The question was attempted by few candidates and the performance was fair.
In(a)(i), candidates correctly stated law of multiple proportions thus:
If two elements, A and B combine to form more than one compound, then the various masses
of one element A which combine separately with a fixed mass of the other element B are in
simple ratio.
In(a)(ii), most candidates failed to correctly calculate and verify the law of multiple proportion
and hence, write the correct formula of P and Q. The expected answers from candidates were
as follows:

= 1.5 x 2 1.0 x 2
= 3 : 2
Since the masses of chlorine which combine with LOg of M in P and Q are in simple ratio 3:2 then
the law of multiple proportion is obeyed
III. P - MCl3
Q - MCl2
In part (b), candidates has shallow knowledge of the IUPAC nomenclature hence could not name
each of the substances in (i) and (ii). The expected answers from candidates were:
(i) Potassium hexacyanoferrate(II);
(ii) Sodium trioxocarbonate (IV) monohydrate.
In(c)(i), candidates correctly stated that salts are compounds formed when all or part of the
ionizable hydrogen of an acid are replaced by metallic or ammonium ions/when all part of the OW
ions of a base are replaced by non metallic ions.
In (cHii), candidates knew that either solubility in water (solubility) or stability to heat as a factor
that influences the method used for the preparation of a salt.
In (cHiii), only few candidates could classify the given salts accordingly. The required answers from
candidates were as follows:
I. Na2Zn(OH) - complex salt,
II. Zn(OH)CI - basic salt;
III. (NH4)2Fe(S04)2.6H20.- double salt.
In(c)(iv), most candidates could not write a two-step equation to show the preparation of sodium
hydrogen trioxocarbonate(lV) from sodium hydroxide. The expected answers from candidates
were: