Question 1
QUESTION 1
a) (i) Write the formula of each of the following compounds:
(ii) Sodium trioxonitrate (V) decahydrate
(iii) Sodium oxide;
(ii) Potassium tetraoxophosphate (V). [3 marks]
(b) Name the products formed when concentrated sodium chloride solution is electrolyzed using graphite electrodes [3 marks]
(c)(i) State the trend in the first ionization energy of the group II elements.
(ii) Explain briefly the answer stated in 1(c)(i). [3 marks]
(d)(i) State the composition of producer gas;
(ii) List two uses of producer gas [5 marks]
(e) State two uses of each of the following compounds
(i) sodium hydroxide;
(ii) sodium trioxocarbonate (IV) [4 marks]
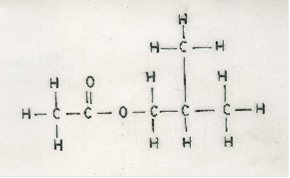
(f) Draw the structure of the main product formed when ethanoic acid reacts with 2-methylpropan-1-ol in the presence of a mineral acid. [2 marks]
(g) State the reason why the isotopes of magnesium, 24Mg and 25Mg, both form ions with +2 charge
12 12 [2 marks]
(h) State one similarity between saponification and neutralization reactions. [1 mark]
(I) When coal is heated in the absence of air, some substances are produced, State the importance of the non-volatile residue to the iron and steel industry. [1 mark]
(j) Give the IUPAC name of the following compound:CH3CH(NH2)CH3 [1 mark]
Observation
This question was compulsory for all candidates. It was reported that higher percentage of the candidates performed below average.
In part (a), majority of the candidates could not write the formulae of sodium trioxonitrate (V) decahydrate, sodium oxide and potassium tetraoxophosphate (V).
In part (b), majority of the candidates could not name the products formed when concentrated sodium chloride solution is electrolyzed using graphite electrodes..
In part (c), only few candidates could state the trend in the first ionization energy of the group II elements.
In part (d), majority of the candidates were able to state the composition of producer gas.
In part (e), majority of the candidates were able to state the uses of sodium hydroxide and sodium trioxocarbonate (IV).
In part (f), majority of the candidates were able to draw the structure of the main product formed when ethanoic acid reacts with 2-methylpropan-1-ol in the presence of a mineral acid.
In part (g), majority of the candidates could not state the reason why the isotopes of magnesium both form ions with +2 charge.
In part (h), majority of the candidates could not state one similarity between saponification and neutralization reactions.
In part (i), majority of the candidates could not state the importance of the non-volatile residue produced when coal is heated in the absence of air to the iron and steel industry.
In part (j), majority of the candidates could not give the IUPAC name of the compound CH3CH(NH2)CH3
The expected answers include:
(a) (i) NaNO3.10H2O
(ii) Na2O
(iii) K3PO4
(b) chlorine gas (at the anode)
hydrogen gas (at the cathode)
sodium hydroxide
(c) (i) First ionization energy decreases down the group II elements.
(ii) Outermost / valence electron further away from the nucleus due to increased shielding / screening (by the inner electrons) / increase in size of atom.
OR
Increase in size outweighs increase in nuclear charge
(d) (i) Producer gas - Carbon (II) oxide/ CO
- Hydrogen/ H2
- Nitrogen/ N2
- Carbon (IV) oxide / CO2
(ii)Uses - fuel
- source of nitrogen
(e)(i) Uses of sodium hydroxide
- used in water treatment
- oil refining
- production of sodium by electrolysis
- manufacture of fabric/textile
- manufacture of soap
- used in paper industry
- purification of bauxite
- as a precipitating agent/analytical reagent
- absorption of CO2
- as oven and drain cleaner
(ii) Uses of sodium trioxocarbonate (IV)
- food processing
- manufacture of glass/paper/soap/detergent/water glass
- softening hardwater
- manufacture of NaOH /borax
- pH modifier
- swimming pool chemical
- as an electrolyte
- standardization of acid/analytical reagent
(g) Both isotopes have the same number of valence electrons / are isoelectronic which are given out
(h) both reactions produce a salt
both reactions require the use of an alkali
both are exothermic reactions
(i) It is used as a reducing agent/smelting/extraction of iron in a blast furnace.
(j) 2 –aminopropane
