Part (a) This question was the least popular among the candidates. The equation was not clearly written by many candidates. Hence the identification of the component was incomplete.
Part(b) Performances by candidates was below average because of ignorance.
Part(c) Performances was above average. The candidates performed very well because they understood the concept very well.
Part(d) Performance was average. However, some candidates, were unaware of the need to consider the relative gaps between the lines. Most candidates indicated correctly the arrow on the transition line, few, forget to insert arrows on the line where necessary because they were ignorant of the concept.
The expected answers are:
12. (a) Einstein’s Photoelectric Equation
E = W + K.Emax
E = Energy of incident radiation
W = Work function
K.E = Maximum kinetic energy of photoelectron
(b) (i) - Emission – frequency/wavelengh of incident radiation
(ii) - Rate of emission - intensity of incident radiation/ number of photons
(iii) - Energy of photoelectrons - frequency/wavelengh of incident radiation
(c) (i) ( 218 + q = 222
q = 4
(β) p = 84 + 2 = 86
m = 214 - 214 = 0
n = 83 – 84 = -1
X is an electron/ beta particle
(ii) Safety Disposal of Radioactive waste
e.g It is - toxic
- emits harmful radiations
(d)(i) E = hf =
= = 8.25 x 10-19
= 8.3 x 10-19J
(ii)
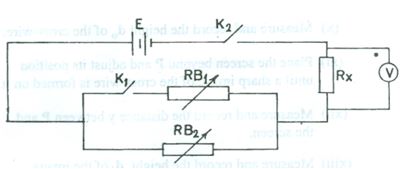
Diagram correctly copied with relative gaps
Arrow correctly shown



