Part (a) was well attempted. However, many of the candidates drew the expected diagram carelessly and so lost valuable marks. One such example is given below:
Part (b) was fairly well attempted.
Part (c) was well attempted by the responding candidates.
The expected answers are:
(a) (i) (A) gamma rays → beta rays → alpha particles.
(B) alpha particles → beta rays → gamma rays.
Accept: γ → β → α and
α → β → γ
(ii)
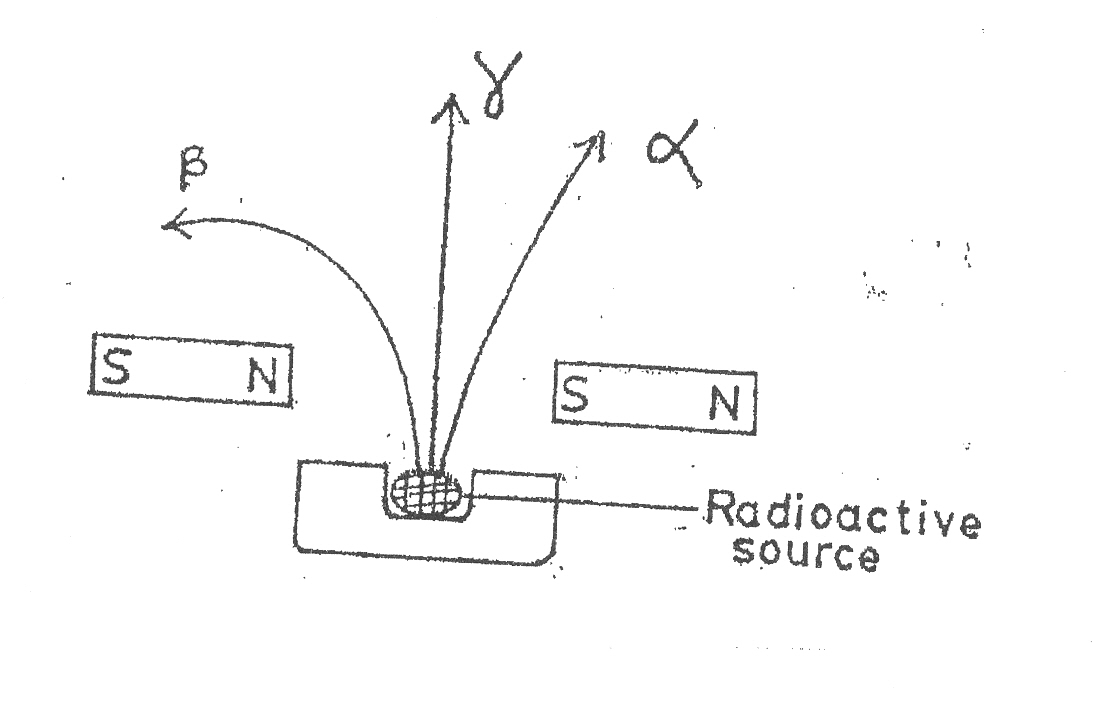
β ray is strongly deflected (because it is negatively charged.
- α particle is deflected but not as strong as the β - beta (because it is massive)
- γ rays is not affected (because it has neither charge nor mass).
(i) - The energy/frequency of incident radiation must be greater than thework function/threshold frequency of the metal
(ii) - the intensity of the radiation
(iii)- Energy/frequency of incident radiation/ work function of the metal irradiated
(c) E = hf
= 6.6 x 10-34 x 5.02 x 1014
= 3.31 x 10-19 J
1 e V = 1.6 X 10-19 J
∴ 3.31 x 10-19 J = 2.07 e V



