Alternative B
Question 2B
You are provided with a lagged calorimeter with a stirrer, a metal block, a measuring cylinder, a thermometer, a retort stand, clamp and other materials.
Use the diagram above as a guide to perform the experiment.
- Measure a volume, v = 50.0 of cold water into the calorimeter, assuming the density of water is 1, determine the mass, of cold water in the calorimeter.
- Measure and record the temperature of the cold water in the calorimeter.
- Place the metal block in the beaker and pour sufficient water to cover the metal in it. Heat the water to boil for at least two minutes.
- Quickly transfer the heated metal into the calorimeter. Stir quickly and record the highest temperature,, of the mixture.
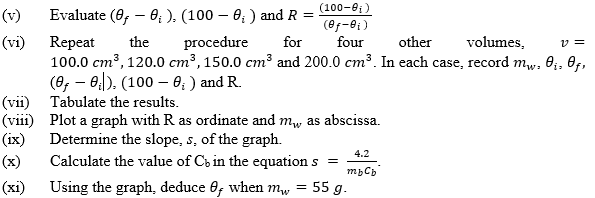 12. State two precautions taken to ensure good results.
12. State two precautions taken to ensure good results.
(b) (i) Explain the statement the specific heat capacity of copper is 385 J Kg-1 K-1.
(ii) A copper ball at 250C is placed in a vessel of water boiling at 1000C until equilibrium is attained. If the mass of the ball is 500g, calculate the thermal energy absorbed by the ball.
[Specific heat capacity of copper = 385 J Kg-1 K-1]
Observation
WEAKNESSES: There is a lack of depth in the concept of heat practicals as most candidates stayed off this question and the few that attempted it did not do well.
STRENGTH: Most candidates explain the statement the specific heat capacity of copper is 385 J Kg-1 K-1 correctly.
EXPECTED RESPONSE:
(a) OBSERVATIONS [07]
- Five values of mw correctly converted to at least 1 d.p in grams.
(Deduct ½ mark for each wrong or missing value)
- Value of θi correctly recorded in °C.
- Five values of θf correctly recorded in °C and in trend.
Trend: As mw increases, θf decreases.
(Deduct ½ mark for each wrong or missing value)
- Five values of (θf - θi) correctly evaluated.
(Deduct ½ mark for each wrong or missing value)
(v) Five values of (100 - θf) correctly evaluated.
(Deduct ½ mark for each wrong or missing value)
(vi) Five values of R = correctly evaluated to at least 2 d.p.
(Deduct ½ mark for each wrong or missing value)
(vii) Composite table showing mw, θf, (θf - θi), (100 - θf) and R.
GRAPH [06]
(i) Both axes correctly distinguished (Award ½ mark each)
(ii) Reasonable Scales (Award ½ mark each)
(iii) Five points correctly plotted
(Deduct ½ mark for each wrong or missing point)
(iv) Line of best fit
SLOPE [02]
(i) Large right-angled triangle
(ii) D R correctly determined
(iii) D mw correctly determined
(iv) correctly evaluated
CALCULATION OF Cb[01]
- Correct substitution
(ii) Correct arithmetic
DEDUCTION OF θf [02]
(iii) Value of R corresponding to mw = 55 g correctly shown on graph.
(ii) , θf correctly calculated.
ACCURACY [01]
Based on candidate’s value of Cb = Teacher’s value of specific heat capacity of
metallic block ± 10%
PRECAUTIONS [02]
Award 1 mark each for any two correct precautions stated in acceptable tense.
Eg:
- Clamped thermometer firmly.
- Avoided splashing of water.
- Avoided parallax error when taking readings on the thermometer.
- Repeated readings(shown on table)
(b) (i) Meaning of statement
385 J of thermal energy is needed to increase the temperature of 1 kg of copper by 1°C/1K
OR
1 kg of copper is required to give out 385 J of thermal energy for its temperature to decrease by 1 °C/1 K.
(ii)
When equilibrium is reached, the temperature of the ball is 100 °C

